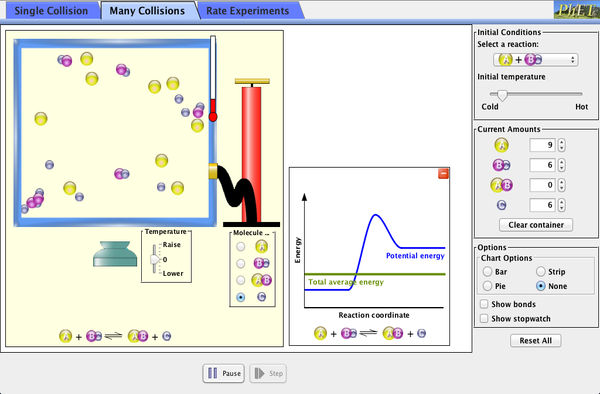
We are starting on the Equilibrium unit this week. Here is a summary of the objectives:
| # | Objective | Must-do Activities | Videos |
| 1 | Explains the state of dynamic equilibrium | Equilibrium Test | Dynamic Equilibrium |
| 2 | Predicts the direction of shift in equilibrium following a change in concentration, temperature, or pressure (Le Châtelier’s Principle) | Le Châtelier assignment, Equilibrium Test | Dynamic Equilibrium, Le Châtelier |
| 3 | Calculates [H3O+] and [OH–], using Kw at 25°C | Kc, Kw, and pH Practice, Equilibrium Test | Kw |
| 4 | Applies the relationship between the pH and the molar concentration of hydronium ions (pH = -log10[H+]) | Kc, Kw, and pH Practice, Equilibrium Test | Calculating pH |
| 5 | Calculates the equilibrium constant (Kc, Ka, Kb) for a chemical reaction | Kc, Kw, and pH Practice, Equilibrium Test | Kc, ICE Tables, Ka, Kb |
| 6 | Associates the strength of acids and bases with the value of their Ka or Kb | Equilibrium Test | Strength vs. Concentration, Ka, Kb |
| 7 | Calculates the Ksp of a substance | Ksp Lab, Equilibrium Test | Ksp |